Our COMPANY

REMY healthcare always puts product quality in the first place. REMY imports goods including all certificates and improves his product quallity from all over the world. From raw material procurement to finished products before leaving factory, REMY healthcare controls quality according to related quality system, so that REMY’s products are in line with the standards of GB, ASTM and EN455 and also have obtained the CE certifications of the EU. To ensure the sustained stability of product quality, REMY has established a quality management system adapting to the production process and imports products with the ISO13485 certification of the TUV.
Our Benefits
MEDICAL MANUFACTURESS
We would like to establish your product in the European Market.
STANDARTS
Our products have the highest medical standards in europe.
CERTIFICATS
We provide you with products having all needed certificates
Office
We provide you and support your company with our Office, Warehouse and Airports
VISIT
We are able to visit your company in days all around the world
24/7 Access
We work all around the WORLD
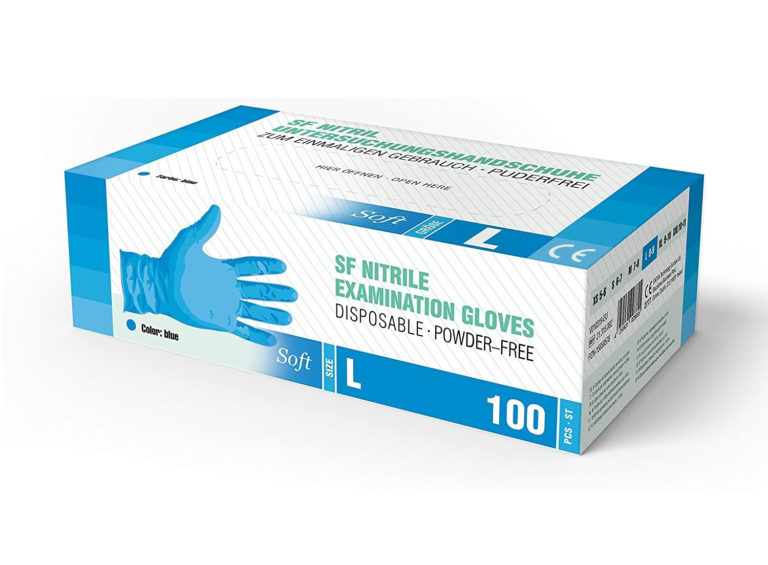
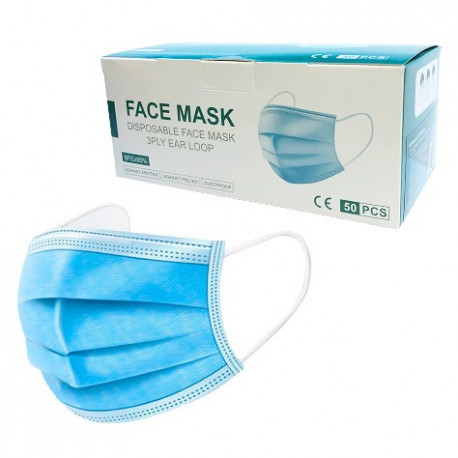
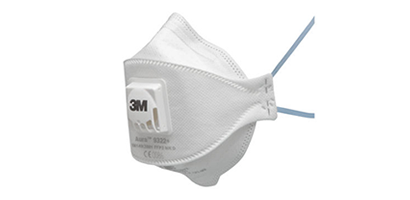
OUR PRODUCTS
F.A.Q.
As a medical device importer, REMY always adheres to the fundamental tenet of “Ensure Safety of Medical Devices, Guarantee Human Health and Life Safety” as well as the business philosophy of “Technology Creates Health, Management Reflects Safety, Quality Casts a Brand”, always attaches importance to the introduction of new technology and new product development and is committed to a green, clean, safe and healthy production method.
REMY healthcare controls quality according to related quality system, so that REMY’s products are in line with the standards of GB, ASTM and EN455 and also have obtained the CE certifications of the EU.
Main OFFICE: REMY, ul. Wróblewskiego. 129 43-210 Kobiór, Poland
WAREHOUSE: REMY, ul. Studzienicka 57, 43-200 Pszczyna, Poland
AIRPORT HOME BASE: – EPKW
We are open for you 24/7.
We are working with
all Mail Service company’s all around the word to deliver our products as fast
as possible to support our clients.
Since there is a high demand of medical products all around Europe we will be proud to bring your products into the European Market.
ALL PRODUCTS NEED TO FULFILL EUROPEEN STANDART NORMS